Development of a Novel Nanosheet with Adjustable Electrical Conductivity
—Unique Hydrogen-Boron Structure Capable of Adsorbing Organic Molecules May Be Applicable to the Development of Molecular Sensors and Catalysts—
2019.12.10
National Institute for Materials Science (NIMS)
University of Tsukuba
Japan Synchrotron Radiation Research Institute (JASRI)
The University of Tokyo
Tokyo Institute of Technology
A research team led by NIMS and University of Tsukuba developed a new nanosheet composed exclusively of born and hydrogen. This research team and JASRI then discovered that the nanosheet contains unique hydrogen atom arrangements which promote molecular adsorption on its surfaces, significantly influencing the electrical conductivity of the nanosheet. This lightweight, flexible nanosheet with adjustable electrical conductivity may apply to the development of wearable electronic devices and sensors with novel working mechanisms.
Abstract
- A research team led by NIMS and University of Tsukuba developed a new nanosheet composed exclusively of born and hydrogen. This research team and JASRI then discovered that the nanosheet contains unique hydrogen atom arrangements which promote molecular adsorption on its surfaces, significantly influencing the electrical conductivity of the nanosheet. This lightweight, flexible nanosheet with adjustable electrical conductivity may apply to the development of wearable electronic devices and sensors with novel working mechanisms.
- Extremely thin (i.e., atomic and molecular-level thickness), electrically conductive nanosheet materials, including graphene, are flexible and have unique electronic states, making them potentially desirable materials for capacitors and other electronic devices. Hydrogen boride nanosheets—composed solely of boron and hydrogen—have been theoretically predicted to have electronic properties superior to those of graphene. Efforts to synthesize this material had been unsuccessful until this research team succeeded in accomplishing it for the first time in the world in 2017. However, after investigating the electronic properties of this nanosheet, the team found it to be electrically non-conductive, contrary to expectations. The team has since been carrying out research to determine the cause of the non-conductivity with the ultimate goal of synthesizing electrically conductive hydrogen boride nanosheets.
- The research team recently discovered that impurities adsorbed on the surface of the nanosheet make it electrically non-conductive. Subsequently, the team succeeded in synthesizing stable conductive hydrogen boride nanosheets by pretreating them to increase their purity. The team then performed nanosheet structural analysis using the SPring-8 synchrotron radiation facility to understand the mechanisms responsible for making the nanosheet electrically conductive or non-conductive. As a result, the team discovered unique hydrogen atom arrangements in the nanosheet which cause electrical unevenness between boron atoms. This promotes the adsorption of small amounts of organic molecules on the nanosheets, destabilizing electrical conduction.
- These results indicate that the electrical conductivity of the hydrogen boride nanosheet may be adjustable by manipulating the adsorption of organic molecules on its surfaces—a potential major advantage. Exploiting this characteristic (i.e., the nanosheet’s ability to adsorb molecules) may make this nanosheet applicable to the development of devices with novel mechanisms, such as molecular sensors and catalysts.
- This project was carried out by a joint research team led by Satoshi Tominaka (Senior Researcher, Soft Chemistry Group, International Center for Materials Nanoarchitectonics, NIMS), Takahiro Kondo (Associate Professor, Faculty of Pure and Applied Sciences, University of Tsukuba), Koji Ohara (Senior Scientist, Center for Synchrotron Radiation Research, JASRI), Iwao Matsuda (Associate Professor, Institute for Solid State Physics, University of Tokyo) and Hideo Hosono (Professor Emeritus, Materials Research Center for Element Strategy, Tokyo Institute of Technology).
- This work was supported by the MEXT Grant-in-Aid for Scientific Research (grant numbers: 18H03874, 18K05192, 19H02551 and 19H05046) and the MEXT Element Strategy Initiative (core research center: Tokodai Institute for Element Strategy).
This research was published in the online version of Chem at 11:00 am on December 9, 2019, local time (1:00 am on December 10, Japan Time).
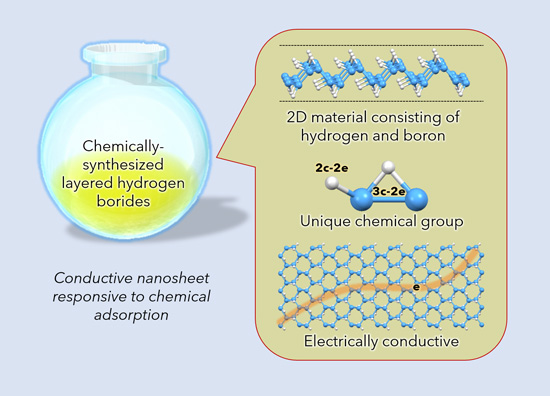
figure: Chemical synthesis of hydrogen boride nanosheets. This molecular-level-thick sheet material has unique hydrogen arrangements and is electrically conductive. Its electrical conductivity is sensitive to the influence of molecular adsorption on its surfaces.
Related files
- MANA
Contacts
(Regarding this research)
-
Satoshi Tominaka
Senior Researcher,
Soft Chemistry Group,
Nano-Material Field,
International Center for Materials Nanoarchitectonics,
National Institute for Materials Science (NIMS)
Tel: +81-29-860-4594
E-Mail: TOMINAKA.Satoshi=nims.go.jp
(Please change "=" to "@") -
Takahiro Kondo
Associate Professor
Faculty of Pure and Applied Sciences
University of Tsukuba
1-1-1 Tennodai, Tsukuba, Ibaraki 305-8577, Japan
Tel: +81-29-853-5934
E-Mail: takahiro=ims.tsukuba.ac.jp
(Please change "=" to "@")
(General information)
-
Public Relations Office
National Institute for Materials Sciences
Tel: +81-29-859-2026
Fax: +81-29-859-2017
E-Mail: pressrelease=ml.nims.go.jp
(Please change "=" to "@") -
Public Relations Office
University of Tsukuba
1-1-1 Tennodai, Tsukuba, Ibaraki 305-8577, Japan
Tel: +81-29-853-2039
Fax: +81-29-853-2014
E-Mail: kohositu=un.tsukuba.ac.jp
(Please change "=" to "@") -
Public Relations Office
Institute for Solid State Physics
The University of Tokyo
5-1-5 Kashiwanoha, Kashiwa, Chiba 277-8581, Japan
Tel: +81-4-7136-3207
E-Mail: press=issp.u-tokyo.ac.jp
(Please change "=" to "@") -
Office of Public Engagement
Public Relations Section
Tokyo Institute of Technology
2-12-1 Ookayama, Meguro-ku, Tokyo 152-8550, Japan
Tel: +81-3-5734-2975
Fax: +81-3-5734-3661
E-Mail: media=jim.titech.ac.jp
(Please change "=" to "@")
(Regarding the SPring-8/SACLA)
-
Information and Outreach Section
User Administration Division
Japan Synchrotron Radiation Research Institute (JASRI)
1-1-1 Kouto, Sayo-cho, Sayo-gun, Hyogo 679-5198, JAPAN
Tel: +81-791-58-2785
Fax: +81-791-58-2786
E-Mail: kouhou=spring8.or.jp
(Please change "=" to "@")
Same Keywords
-
High-speed switching for ultrafast electromechanical switches and sensors
(SPring-8,JASRI,Tokyo Institute of Technology)
2017.08.29
-
Conventional Idea of Electronic State in High-Temperature Superconductors Is Overturned
(SPring-8,JASRI)
2021.04.13
-
Discovery of Nanosheets with the Highest Ever Hydroxyl Ion Conductivity
(conductivity,nanosheet)
2017.04.15
