Nanoparticle Group
Nanomaterials Field
We develop novel colloidal nanocrystals as part of cutting-edge materials for optoelectronic and bio-applications
Group Leader:Naoto Shirahata
Computationally guided defect-suppressing synthesis for highly luminescent tin halide perovskite nanocrystals
Current Topics
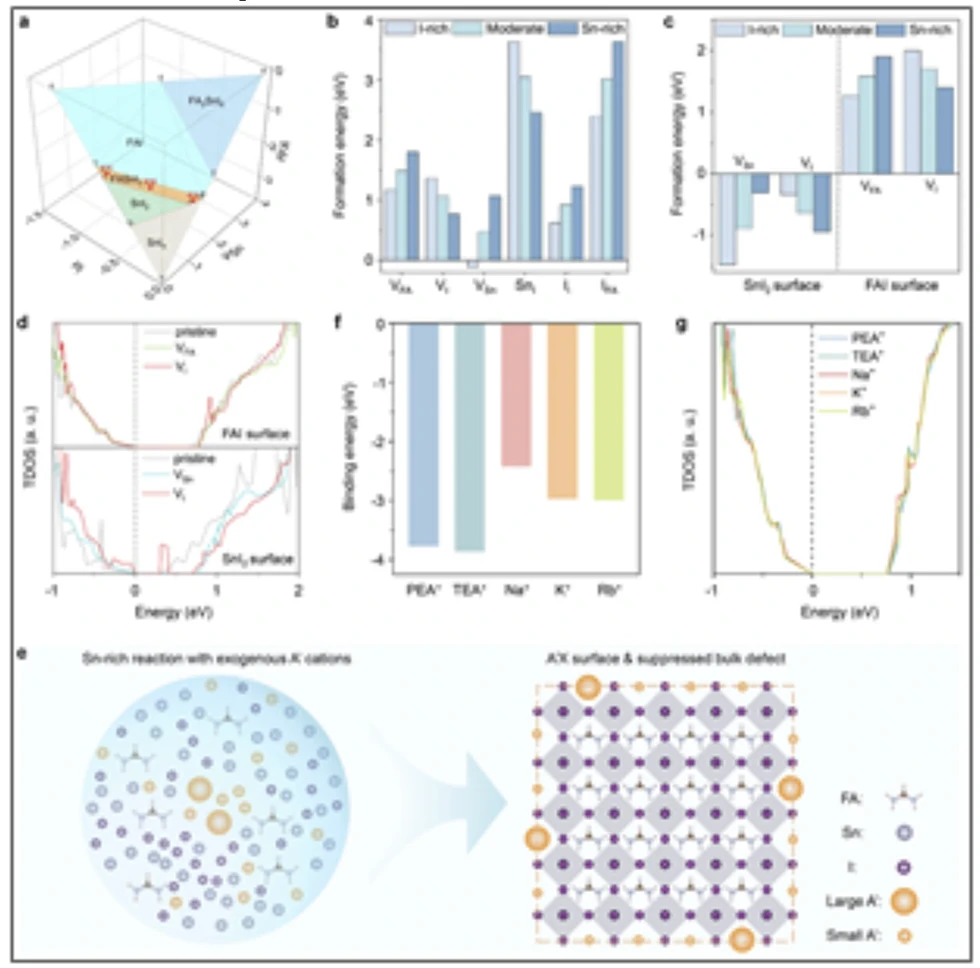
Colloidal lead halide perovskite nanocrystals exhibit photophysical properties that include high photoluminescence quantum yield, excellent color purity, tunable emission from visible to near-infrared, and large absorption cross-section. These attributes have made them promising for a range of optoelectronic applications. However, reliance on lead raises regulatory concerns, motivating the search for lead-free alternatives. Tin halide perovskite nanocrystals are candidate lead-free alternatives for optoelectronic applications. However, their synthesis remains challenging due to limited understanding of defect chemistry and the lack of defect-suppression strategies. This is reflected in the low photoluminescence quantum yields of tin perovskite nanocrystals synthesized via protocols optimized for lead counterparts.
To overcome this challenge, we present a computationally guided approach that informs the synthesis of hybrid tin perovskite nanocrystals. Calculations reveal that the formation of structural defects in FASnI3 (FA = formamidinium) is governed by the chemical potentials of the constituent (FA, Sn, and I) precursors: Sn-rich conditions suppress bulk defects, whereas Sn-poor conditions reduce surface defects by forming defect-tolerant FAI-terminated surfaces (Figure 1a-d). This indicates that dual defect suppression is not readily realized using only constituent precursors. We therefore introduce exogenous monovalent cations that do not generate deep trap states, enabling the formation of defect-tolerant surfaces under Sn-rich conditions while independently tuning precursor ratios to optimize the chemical potentials (Figure 1e-g). This strategy leads to FASnI3 nanocrystals with a record photoluminescence quantum yield of 42.4±1.0%. We demonstrate that this strategy can be extended to other tin perovskite NCs such as FA/Cs alloyed tin perovskite nanocrystals.
References
Group members
-

Naoto Shirahata
- Group Leader
-

Hong-Tao Sun
- Principal Researcher
Links
-
Semiconductor Materials Field - Nanostructured Semiconducting Materials Group
- Quantum Materials Simulation Group
- Semiconductor Functional Device Group
- Thin Film Electronics Group
- Neuromorphic Devices Group
- Semiconductor Nano-integration Group
- 2D Semiconductor Group
- Photonics Nano-Engineering Group
- Ionic Devices Group
- Semiconductor Device Group
- Smart Interface Team
-

Quantum Materials Field -
Nanomaterials Field - Thermal Energy Materials Group
- Soft Chemistry Group
- Functional Nanomaterials Group
- Layered Nanochemistry Group
- Frontier Molecules Group
- Functional Chromophores Group
- Supermolecules Group
- Nanoparticle Group
- High-Pressure Structural Controls Group
- ElectroActive Materials Team
- Optical Nanostructure Team
-

Independent Researcher
Top


