Battery Materials Analysis Group
Members
Takuya MASUDA
SAMURAI
Director, Research Center for Energy and Environmental Materials (GREEN)
Team Leader, Advanced Characterization Team, Center for Advanced Battery Collaboration, Research Center for Energy and Environmental Materials (GREEN)

Hidenori NOGUCHI
SAMURAI
Chief Researcher, Battery Materials Analysis Group, Battery and Cell Materials Field, Research Center for Energy and Environmental Materials (GREEN)

Kunie ISHIOKA
SAMURAI
Principal Researcher, Battery Materials Analysis Group, Battery and Cell Materials Field, Research Center for Energy and Environmental Materials (GREEN)

Kimihiko ITO
SAMURAI
Principal Researcher, Battery Materials Analysis Group, Battery and Cell Materials Field, Research Center for Energy and Environmental Materials (GREEN)

Kei KUBOTA
SAMURAI
Senior Researcher, Battery Materials Analysis Group, Battery and Cell Materials Field, Research Center for Energy and Environmental Materials (GREEN)

Hisao KIUCHI
SAMURAI
Senior Researcher, Battery Materials Analysis Group, Battery and Cell Materials Field, Research Center for Energy and Environmental Materials (GREEN)

Motivation and Outline
Practical rechargeable batteries are densely packed with various materials that undergo complex physical and chemical processes during charging and discharging.
These processes include the electrochemical reactions of active materials, changes in crystal structure, plastic deformation, stress concentration, and crack formation.
Our research focuses on developing various in situ/operando techniques to observe the complex physicochemical phenomena underlying battery characteristics and clarify their quantitative impact on macroscopic battery performance.
Our goal is to establish the fundamental principles necessary for designing high-performance, long-lasting battery materials.
Facilities
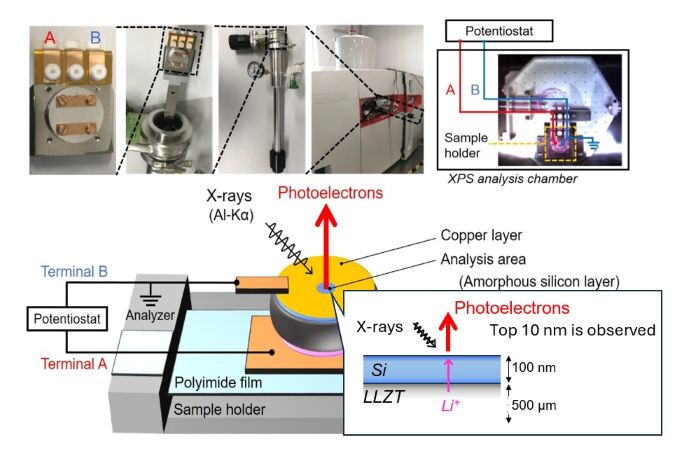
X-ray photoelectron spectroscopy apparatus (AXIS NOVA, SHIMADZU Kratos)
Raimu Endo, et al., J. Phys. Commun., 5 (2021) 015001
Raimu Endo, et al., J. Phys. Chem. Lett., 11 (2020) 6649-6654
Raimu Endo, et al., J. Phys. Chem. Letters. 13 [31] (2022) 7363-7370

Atomic Force Microscopy (Cypher ES, Asylum Research, Oxford Instruments)
Hiroki Sakai, et al., J. Power Sources., 413 (2019) 29-33.
Ridwan P. Putra, et al., J. Phys. Chem. Lett., 15 (2024) 490–498.

Vibrational Sum Frequency Generation Spectroscopy System
(EKSPLA:PL2251A-20+PG501-DFG)
Mikio Ito, et al., J. Chem. Phys., 161 (2024) 204703
Naoto Ogiwara, et al., ACS Appl. Mater. Interface, 16 (2024) 44575-44589
Moataz Mekawy, et al., J Colloid Interf. Sci., 613 (2022) 311-319
Research Results
Using a bimodal mode atomic force microscope (AFM) system, we performed nanomechanical mapping of amorphous Si thin film electrodes during the lithium insertion and extraction process. We successfully observed the Young's modulus distribution and surface morphology of the Si electrode in real time, depending on the amount of lithium (x) in lithium silicide (LiₓSi). During the initial stage of lithium insertion, the Young's modulus decreased sharply due to the transformation from Si to LiₓSi, and then decreased gradually until x = 3.46 (capacity 3300 mAh/g), enabling us to establish a correlation between the lithium content (x) and the Young's modulus. Applications to various battery materials are on-going.
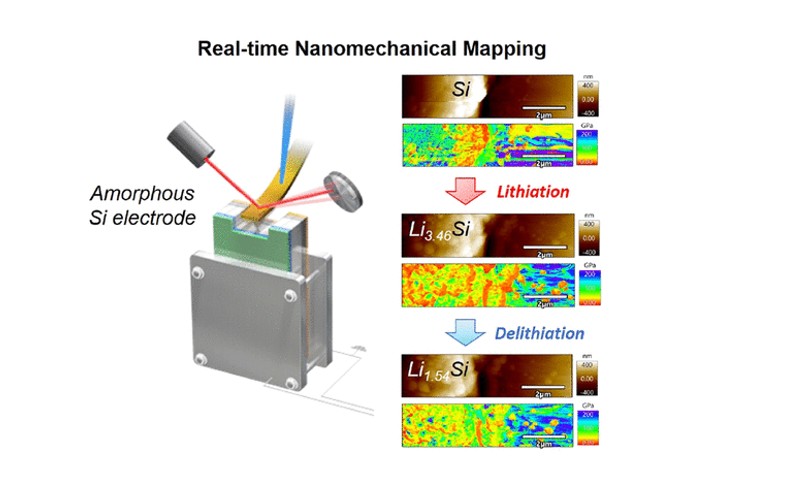
Ridwan P. Putra, et al., J. Phys. Chem. Lett., 15 (2024) 490–498.



