Research
We are devoted to the research of biodegradable scaffolds and functional biomaterials for tissue engineering, cancer therapy and regulation of cell functions. Porous scaffolds with well-controlled pore structures, composite scaffolds of biodegradable synthetic polymers and naturally derived polymers, multi-functional photothermal scaffolds for simultaneous applications of cancer therapy and tissue engineering, and highly biocompatible matrix biomaterials with nano- and microstructures constructed from cultured cells are designed and prepared. Biomaterials that mimic the in vivo nano- and microenvironments, cell-encapsulation hydrogels, and micro-patterned surfaces are created to manipulate cell functions, particularly stem cell functions.
1.Development of Tissue Engineering Scaffolds
Scaffolds play an important role as a temporary support for accommodation of seeded cells to control cell functions and to guide regeneration of functional tissues and organs. In addition to controlling cell functions, scaffolds should have good biocompatibility, degradation, favorable pore structures and high mechanical strength. We have prepared a variety of porous scaffolds of biodegradable polymers. We have hybridized biodegradable synthetic polymers and naturally-derived polymers to prepare composite scaffolds that combine the advantages of both types of biodegradable polymers. We have developed a method using pre-prepared ice particulates as templates to precisely control the porous structures of scaffolds. Scaffolds with micropatterned pore structures have been prepared by using embossing ice particulates of different micropatterns. These scaffolds have been used for tissue engineering of skin, bone, cartilage, muscle and etc.
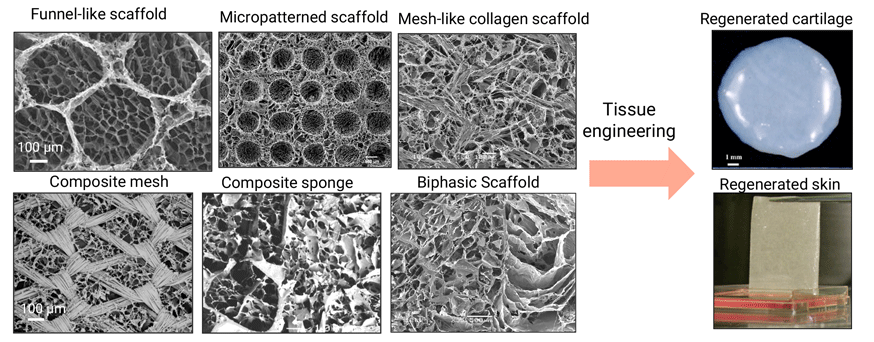
2.Development of Functional Composite Scaffolds for both Cancer Therapy and Tissue Engineering
Eradication of remaining and metastatic cancer cells to protect cancer recurrence remains a challenge for cancer therapy. Meanwhile regeneration of large defect after surgical resection is desirable for improving the quality of life of patients. We have prepared multi-functional composite scaffolds for both cancer therapy and tissue engineering. Photothermal conversion nanoparticles such as gold nanoparticles, black phosphorus (BP) nanosheets and magnetic iron oxide nanoparticles have been hybridized with biodegradable polymers such as collagen and gelatin to prepare the composite scaffolds. The composite scaffolds can generate heat to ablate cancer cells under irradiation of near infrared laser or alternating magnetic field, and subsequently induce tissue regeneration to heal the defects after ablation of cancer cells.

3.Preparation of Biomimetic Extracellular Matrix Scaffolds
The extracellular matrix (ECM) interacts with cells and can provide various signals for controlling cell functions and maintaining the homeostasis of living organisms. The composition of ECM is not only dependent on the type of tissues and organs but is also dynamically remodeled and balanced during the development and aging process. We have used cell culture technology and decellularization to prepare biomimetic matrices and ECM scaffolds. Stepwise tissue development-mimicking matrices and scaffolds, and autologous scaffolds have been prepared by the method and used for controlling stem cell functions and tissue engineering.

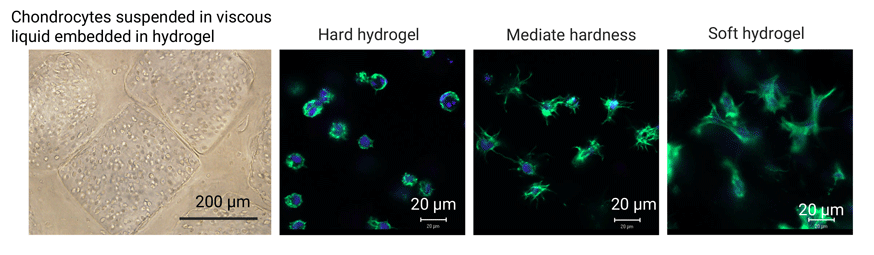
4.Synthesis of hydrogels for 3D cell culture
Cells in each tissue and organ sense and respond to changes in their microenvironments to maintain their metabolic activity and phenotypes. The stiffness and viscoelastic properties of the extracellular matrix have significant modulatory effects on cell functions such as cell adhesion, morphology, mobility, self-renewal, proliferation and differentiation. We have established a 3D culture system of hydrogels to investigate the influence of hydrogel stiffness and liquid viscosity on cell functions. Photocrosslinkable gelatin methacryloyl (GelMA) macromers are synthesized by introducing methacryloyl groups in gelatin molecules. Cells are encapsulated in the GelMA hydrogels to investigate the influence of hydrogel stiffness on cell functions. Cell-suspended viscous liquid droplets are embedded in the hydrogels to elucidate the impact of liquid viscosity on cell functions.
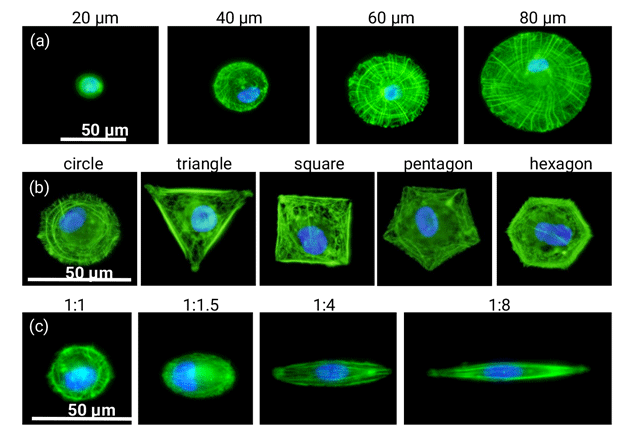
5.Preparation of micropatterns for regulation of cell functions
Manipulation of stem cell functions is important for tissue engineering. Cells exist in their respective in vivo microenvironments that are important for the maintenance of cell functions and metabolism. The microenvironments include biological and physicochemical cues to dictate cell migration, adhesion, proliferation, differentiation and secretion of extracellular matrix secretion. Biological cues such as cell-cell interaction and cell-ECM interaction, and physicochemical cues such as chemical moiety, electrostatic properties, size, shape, stiffness, roughness and topography have been widely studied to elucidate their influence on cell functions, especially on differentiation of stem cells. We have prepared a number of micro-patterned surfaces by photolithography through designing the photomask features. Cell spreading area, geometry, aspect ratio, alignment, chirality and cell-cell interaction have been regulated by the micro-patterned structures to investigate their influence on cytoskeleton organization, cell mechanics, stemness, differentiation and gene transfection of stem cells.