- ホーム
- > 研究活動
- > Research Highlights
- > Vol. 49 Porous Structure of a Layered ・・・
 Research Highlights
Research Highlights
[Vol. 49]
Porous Structure of a Layered Silicate with Selective Adsorption Properties Revealed
- 前の記事
- 一覧に戻る
- 次の記事
2019.03.20
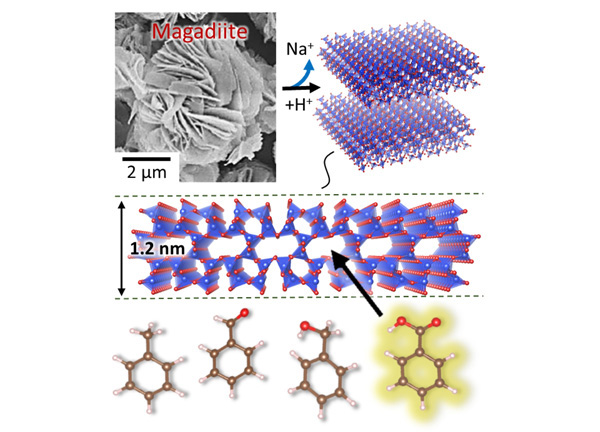
Figure: The structure of magadiite features pores that have a selective adsorption function.
It is not always easy to precisely determine the crystal structure of layered materials — but in order to fully understand and exploit their properties, detailed structural information is needed. Magadiite is such a layered material, used as an adsorbent and a catalyst. It is known that magadiite is a kind of layered silicates: tetrahedra, each with oxygen atoms at the vertices and a silicon atom in the center, grouped in planar arrangements. The precise structure, however, was not known — partly because the material typically occurs as small lamellas displaying poor crystallinity — until now. Satoshi Tominaka at the International Center for Materials Nanoarchitectonics (MANA), National Institute for Materials Science (NIMS), Tsukuba, Japan, and colleagues have succeeded in determining the crystal structure of magadiite. Based on their structural insights, Yusuke Ide (MANA, NIMS) and colleagues also managed to explain why the material has outstanding photocatalytic properties and proved its application in the synthesis of pure benzoic acid from toluene using a photocatalyst system.
The researchers looked at Na-magadiite (the natural, sodium-containing form) and H-magadiite (the ‘protonated’ form, obtained by removing sodium). By collecting X-ray diffraction data and using a method called pair-distribution-function analysis, they were able to come up with a structural model. Importantly, they found that in Na-magadiite, along the silicate layers, micropores filled with sodium atoms occur. Removing the Na content (by acid treatment) resulted in a structure with channels.
The porosity of H-magadiite was confirmed by looking at nitrogen gas (N2) adsorption. N2 molecules can enter the pores; the size of the pores was estimated to be around half a nanometre.
The team also found that although the pores in H-magadiite are small, they can absorb benzoic acid (C6H5COOH). This property is of high importance in the photocatalytic synthesis of benzoic acid from toluene (C6H5CH3), with titanium oxide (TiO2) as a catalyst. By letting the synthesis take place in an environment with H-magadiite, the yield of benzoic acid was much higher than normal; the explanation is that the pores of H-magadiite temporarily store the C6H5COOH molecules.
Now that the full structure of magadiite has been established — including the geometry of the micropores along the thin silicate layers — further applications of the material can be investigated. Quoting the scientists: “These structural features of magadiite are expected to create advanced applications of magadiite as we demonstrated [for the case of benzoic acid production with magadiite as a selective adsorbent].”
Reference
“Zeolitic intralayer microchannels of magadiite, a natural layered silicate, to boost green organic synthesis”
Yusuke Ide, Satoshi Tominaka, Hiroyuki Kono, Rahul Ram, Akihiko Machida, and Nao Tsunoji
Journal : Chem. Sci. 9 8637 (2018).
DOI : 10.1039/c8sc03712d
Yusuke Ide, Satoshi Tominaka, Hiroyuki Kono, Rahul Ram, Akihiko Machida, and Nao Tsunoji
Journal : Chem. Sci. 9 8637 (2018).
DOI : 10.1039/c8sc03712d
Affiliations
International Center for Materials Nanoarchitectonics (WPI-MANA), National Institute for Materials Science (NIMS), Namiki 1-1, Tsukuba, Ibaraki 305-0044, Japan
Contact information
ナノアーキテクトニクス材料研究センター (MANA)
〒305-0044 茨城県つくば市並木1-1
TEL: 029-860-4710
E-mail: mana-pr=ml.nims.go.jp([ = ] → [ @ ] )
TEL: 029-860-4710
E-mail: mana-pr=ml.nims.go.jp([ = ] → [ @ ] )