 Press Release 2017
Press Release 2017
Discovery of Nanosheets with the Highest Ever Hydroxyl Ion Conductivity
- Technology May Contribute to the Realization of Highly Efficient Solid Alkaline Fuel Cells -
A NIMS research group discovered that layered double hydroxide nanosheets have exceptionally high hydroxyl ion conductivity (as high as 10-1 S/cm).
(“Single-layer nanosheets with exceptionally high and anisotropic hydroxyl ion conductivity”, Pengzhan Sun, Renzhi Ma, Xueyin Bai, Kunlin Wang, Hongwei Zhu, and Takayoshi Sasaki, Science Advances Vol. 3, no. 4, e1602629, DOI: 10.1126/sciadv.1602629)
In fuel cells, which are drawing attention as a clean energy conversion technology, hydrogen ion (H+) conductors (e.g., Nafion®) are usually used as electrolytes. However, the use of H+ conductors virtually requires the use of platinum-based catalysts, because H+ creates a highly acidic operating environment. It is feasible to employ OH- in place of H+ as the conducting ion. When OH- is used, the operating environment is alkaline, allowing the use of other cheaper transition metal elements, such as Fe, Co and Ni, as catalysts, reducing production costs. The major issue with this approach, however, is that the conductivity of OH- in existing OH- conductors is low (10-3 to 10-2 S/cm). Demand has been high to develop practical conductor materials with ionic conductivity of about 10-1 S/cm, which is comparable with the conductivity of H+ conductors.
In this study, the research group exfoliated LDHs into single layers in chemical reactions, and measured the ionic conductivity of the resulting single-layer nanosheets. The nanosheets demonstrated very high conductivities, up to 10-1 S/cm, at approximately room temperature. The high conductivity may be explained as follows. A large amount of moisture adsorbs onto the surface of single-layer nanosheets, promoting OH- to move freely on the surface, thereby dramatically enhancing the nanosheets’ ion transport properties. The conductivity attained in this study is higher than that of any other OH- conductor reported previously. In addition, conductivity in a direction parallel to the nanosheet surface (in-plane direction) was four to five orders of magnitude higher than conductivity in a direction perpendicular to the surface (cross-plane direction). Therefore, the high conductivity observed may be attributed to the ultimate two-dimensional nanostructure of the sheets.
This study’s findings may serve as a major step toward the realization of solid fuel cells driven by OH-, which have been anticipated for many years. In order to apply the superior in-plane ionic conductivity identified in this study to solid electrolyte layers for fuel cells and water electrolyzers, it will be vital to design device structures capable of fully exploiting said conductivity.
Part of this study was carried out in conjunction with a project entitled “Function tuning by means of morphology and structure control of low-dimensional hydroxide nanostructures,” funded by the MEXT Grants-in-Aid for Scientific Research (B).
This study was published in the online version of the US scientific journal Science Advances on April 14, 2017. (DOI: 10.1126/sciadv.1602629)
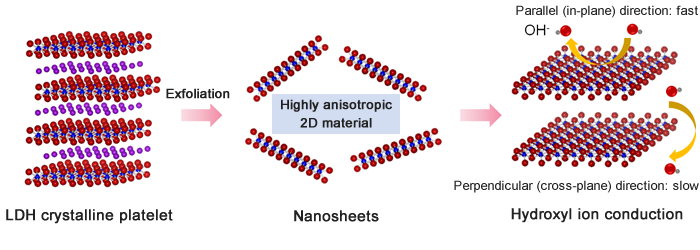
Figure: A layered double hydroxide (LDH) crystalline platelet was exfoliated into single-layer nanosheets—the minimum basic units—which are highly anisotropic in terms of ionic conductivity.
(Regarding this research)
Renzhi Ma
Associate principal investigator, Soft Chemistry Group, MANA, NIMS
Tel:+81-29-860-4124
E-Mail:MA.Renzhi=nims.go.jp
(Please change "=" to "@")
Takayoshi Sasaki
Director, MANA, NIMS
Tel:+81-29-860-4313
E-Mail:SASAKI.Takayoshi=nims.go.jp
(Please change "=" to "@")
(For general inquiries)
Public Relations Office
National Institute for Materials Science
1-2-1 Sengen, Tsukuba, Ibaraki, 305-0047, JAPAN
Tel:+81-29-859-2026
Fax:+81-29-859-2017
E-Mail:pressrelease=ml.nims.go.jp
(Please change "=" to "@")

